ARCHEOLOG-HOME / INDIana-UNIversitas
Infants' Flexible Heads Stretch Back Millions of Years
Infants' Flexible Heads Stretch Back Millions of Years
Michael Balter
Source - http://news.sciencemag.org/sciencenow/2012/05/infants-flexible-heads-stretch.html
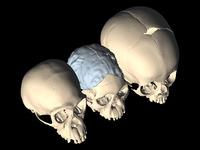
Looking for closure. By comparing CT scans of a 3-million-year-old hominin (center) with chimps (left) and modern humans (right), researchers found that its skull sutures fuse relatively late like today's humans. Credit: M. Ponce de León and C. Zollikofer/University of Zurich
It isn't easy being born. Human babies have big heads, which makes their passage through the birth canal a challenge for both them and their mothers. Fortunately, an infant's skull can change shape as it squeezes through because its cranial bones don't entirely fuse together for at least 2 years after birth. A new study shows that this delayed fusion was also a feature of early humans who lived nearly 3 million years ago, even though their heads were much smaller than ours. One possible explanation is that walking upright created new obstetrical challenges even for small-brained human ancestors.
For chimpanzees and other primates, childbirth is relatively easy. The brain of a newborn chimp is roughly 155 cubic centimeters (cc) in volume, less than half that of a newborn human baby, although the overall size of the birth canal is about the same as in humans. Yet there is a big difference in shape between chimp and human pelvises. While the human pelvis has widened considerably over the course of evolution, the demands of upright walking have put constraints on how wide it can be. Bipedalism has also led to a marked vertical shortening of the pelvis, leading to what researchers call the "obstetric dilemma"—the difficult tradeoff between the demands of bipedalism and having babies.
Still, thanks to those unfused skull bones, the baby's head can not only be molded to fit through the pelvic canal, but can also accommodate the explosive brain growth that takes place after birth: In the first few years of life, the human brain doubles in size from about 400 cc to 800 cc, and ultimately reaches an adult average of about 1400 cc.
To see how far back in human evolution this delayed fusion goes, a team led by anthropologist Dean Falk of the School for Advanced Research in Santa Fe, New Mexico, looked at a key marker of skull fusion in a large number of fossil early humans, modern humans, chimpanzees, and bonobos. The centerpiece of the study was a new analysis of the nearly 3-million-year-old Taung child, an australopithecine roughly 4 years old at death discovered in South Africa in 1924 by the legendary anthropologist Raymond Dart. The specimen, which Dart assigned to the species Australopithecus africanus, includes a face, lower jaw, and a natural "endocast" of the skull made up of the rocky material that filled it. The endocast preserved many of the skull's features, including the sutures between its bones.
Falk, together with anthropologists Christoph Zollikofer and Marcia Ponce de León of the University of Zurich in Switzerland, used computerize tomography scans of the Taung endocast to closely examine the child's metopic suture (MS), which forms the joint between the cranium's two frontal bones. In human children, the MS begins to fuse on the end closest to the nose, closing like a zipper until it reaches the anterior fontanelle, the baby's "soft spot" (see illustration). The team found that the Taung child's MS was unfused, even though its brain was only about 400 cc and the brain of adult A. africanus is only about 460 cc.
The researchers compared the Taung child's MS to that of several hundred chimps and bonobos, more than 1000 modern humans, and 62 hominins, or ancient humans, including australopithecines, Homo erectus, and Neandertals. A clear pattern emerged: The MS of chimps and bonobos fuses very shortly after birth; whereas, like the Taung child, the MS of both early and later hominins tends to fuse only after the eruption of the first molars, at 2 years of age or later. The results are reported online this week in the Proceedings of the National Academy of Sciences (PNAS).
So why do the skulls of early, small-brained hominins already show the fusion patterns of later, big-brained species? Falk and her colleagues propose three possibilities. First is the "obstetric dilemma," which may have already been a problem for early hominins as they began to walk upright. Thus the brain of an adult A. africanus, while small by modern human standards, is still about 22% larger than that of a chimp. Second, the team suggests, the delayed fusion might have been necessary for accelerated brain growth after birth, which might already have been a feature in early hominins. Finally, changes in the organization of the australopithecine brain, such as an apparent widening of the frontal lobes, might also have made the trip through the birth canal more difficult, even if the overall size of the brain remained relatively small.
"All of these are strong possibilities," says Owen Lovejoy, an anthropologist at Kent State University in Kent, Ohio, who edited the paper for PNAS. "There is now reasonable evidence that [australopithecine] brains were to some degree enlarged over those of living chimpanzees, and it is reasonably certain that they were reorganized."
But Robert Martin, an anthropologist at the Field Museum in Chicago, Illinois, urges caution about interpreting the results. He says that the team has placed too much emphasis on both the Taung child endocast as its key specimen and the MS itself, whose age of closure is "notoriously variable" in modern humans. Martin thinks the team would have done better to concentrate on the closure of the anterior fontanelle "soft spot," which he says might be a more reliable indicator of when overall skull fusion takes place.